AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Native dot blot12/4/2023  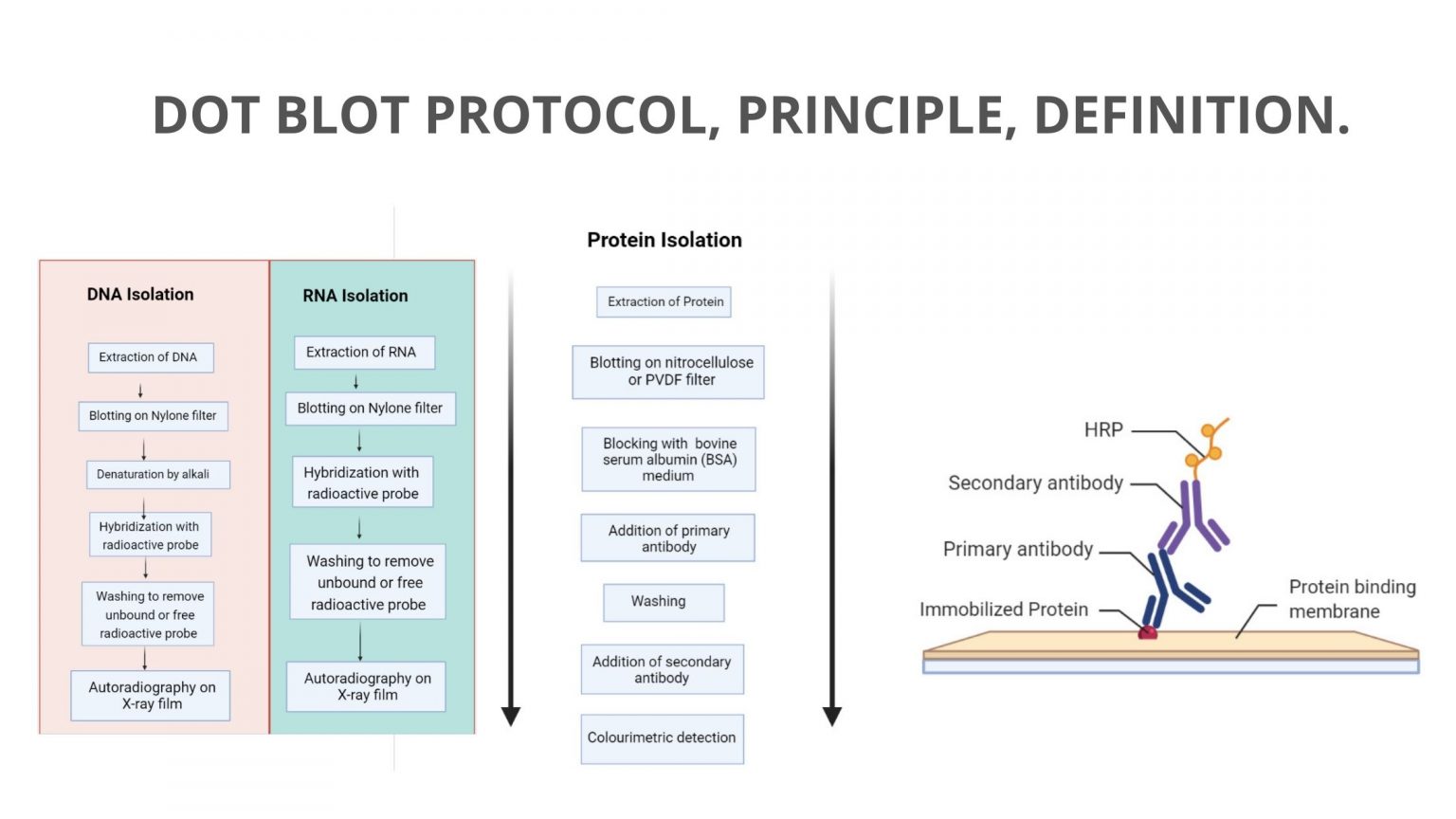
Wash the membrane 2X for 5 minutes each with Wash Buffer.E.g 5 minutes for EveryBlot Blocking Buffer). Rinse the membrane twice with PBS and incubate with Blocking Buffer at room temperature for 1 hour with rocking (If chemical blocking buffer is used, follow manufacturer recommendation for blocking time.Note: Use a low-fluorescence PVDF membrane or nitrocellulose membrane for fluorescence imaging. If using wet transfer, use 100 V for 60 minutes.If using semi-dry transfer, use 10 V for 30 minutes.Assemble transfer sandwich as per instrument instruction and then transfer the proteins onto a PVDF membrane.Incubate gel in cold Transfer Buffer for 15 minutes. 35 µL) of samples per well in a 10% polyacrylamide gel. Heat the samples at 95☌ for 5 minutes.In this case, it is recommended to run a small volume of the Positive Selection Cocktail to check for background signals. If you used EasySep™ to isolate the EVs, the antibodies from EasySep™ Positive Selection Cocktail may be detected by secondary antibodies.To prepare 4X Laemmli Sample Buffer with reducing agent, add 100 µL of 2-mercaptoethanol to 900 µL of 4X Laemmli Sample Buffer, or add dithiothreitol to 4X Laemmli Sample Buffer to a final concentration of 50 mM. When uncertain, test 4X Laemmli Sample Buffer with and without reducing reagent. EV markers other than CD9/CD63/CD81/CD45/EpCAM may require a reducing agent added to Laemmli Sample Buffer.Note: When blotting for CD9, CD63, CD81, EpCAM, and CD45, do not add a reducing agent to the Laemmli Sample Buffer, as CD9/CD63/CD81 detection antibodies often recognize the disulfide bond on the antigen's epitope. Here, we provide a detailed protocol for performing a western blot to detect the presence of such characteristic EV-associated proteins in order to confirm the presence of EVs in the biological sample. Tetraspanin markers such as CD9, CD63, and CD81 are proteins commonly found on EVs across different cell types. Isolated EVs can be characterized by western blotting (also referred to as immunoblotting), a widely used technique to detect specific protein markers in a sample. Once isolated, the particles should be analyzed to confirm that they are indeed EVs and not products of cell fragmentation or other contaminants such as protein complexes or lipoproteins, usually present in biological samples. Using EasySep™ Human Extracellular Vesicle Positive Selection Kits or Extracellular Vesicle SEC Columns, researchers can easily isolate and purify human EVs from biofluids-including serum and plasma-and from culture-conditioned media. MesenCult™-ACF Plus Medium), such as immunomagnetic separation, differential ultracentrifugation, or size exclusion chromatography (SEC)*. There are several ways for isolating EVs from biofluids or cell culture conditioned media (e.g. Due to their inherently heterogeneous nature as well as the complexity of biological samples, it is recommended to characterize EVs after isolation. These vesicles, carrying protein and genetic cargo, play an important role in intercellular communication and are recognized for their potential therapeutic applications. Tissue and Cell Culture Dissociation ReagentsĮxtracellular vesicles (EVs) are lipid bilayer-enclosed structures released by almost all cell types.The biofabricated auxetic scaffold represents an excellent platform for exploring interactions between cells and their mechanical microenvironment.Work at STEMCELL View Current Opportunities > The function of HC was influenced by cyclic tensile stimulation, as shown by increased production of the chondrogenesis-related markers, collagen II and glycosaminoglycans, with the involvement of the yes-associated protein 1 signaling pathway. We show that when human chondrocytes (HCs) were incorporated into these scaffolds, their proliferation and the expression of chondrogenesis-related markers increased with dECM content. Different concentrations of decellularized extracellular matrix (dECM) were mixed with fish gelatin methacrylate (FGelMa) and employed for the preparation of dECM/FGelMa auxetic bio-scaffolds using 3D biofabrication technology. In this study, we fabricated an auxetic scaffold to investigate the effect of 3D tensile stimulation on chondrocyte behavior. Various studies have shown that mechanical cues affect the cellular microenvironment and thereby influence cellular behavior. Three-dimensional (3D) hydrogel constructs can mimic features of the extracellular matrix (ECM) and have tailorable physicochemical properties to support and maintain the regeneration of articular cartilage. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
